The ZECA concept: The process is shown as a block diagram below. It involves the gasification of coal in H2 (hydrogasification) and is designed to avoid using O2 for combustion in the provision of process energy. This integrated process route has not been studied experimentally, although different parts of the concept have been tested (e.g. hydrogasification and CaO driven steam reforming/shifting of CH4). However, an independent study by Nexant Inc. has concluded that the concept is potentially viable and efficient, but that experimental studies are needed to determine reaction conditions, prior to developing a detailed process flow sheet. 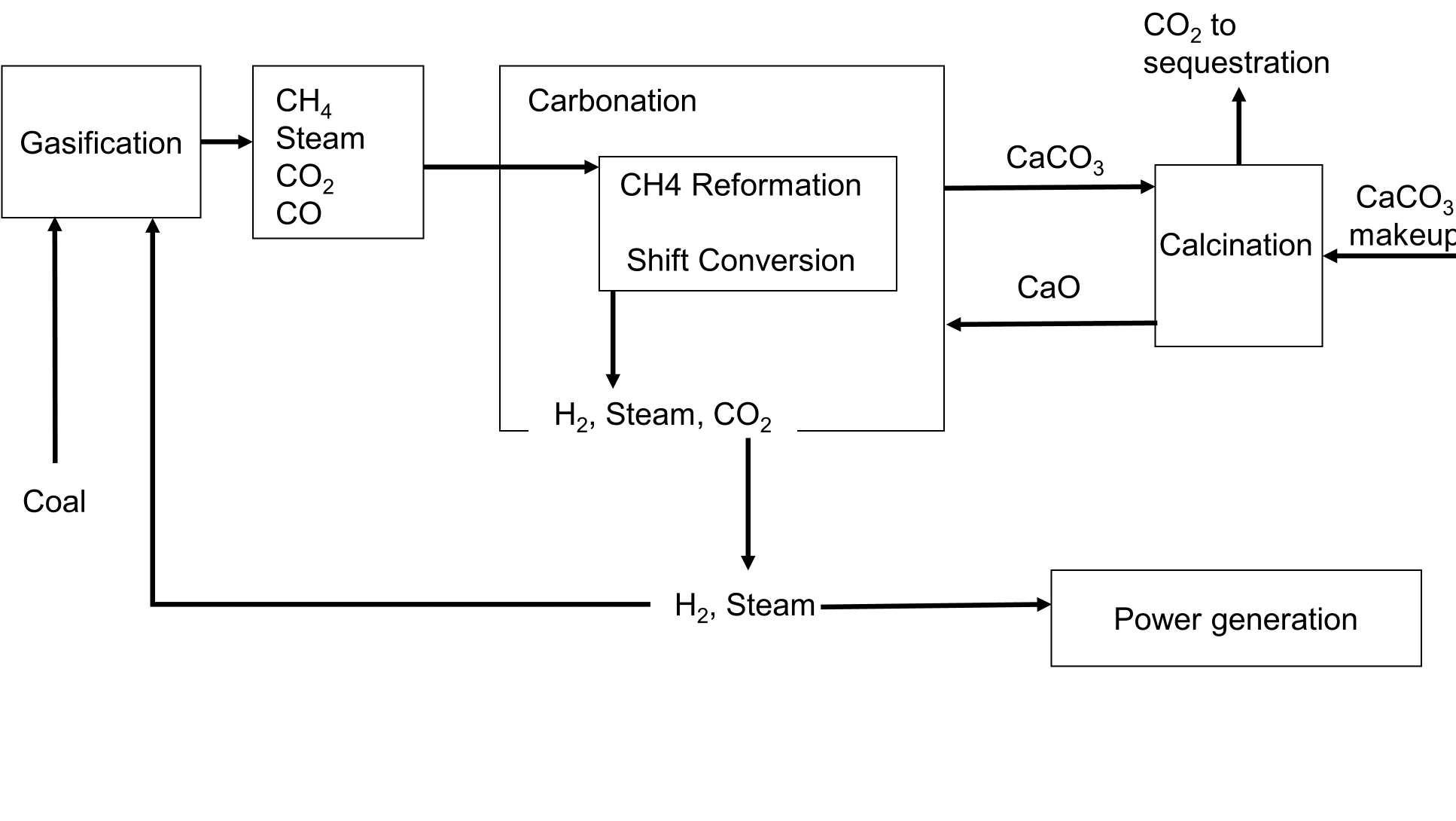
Coal is gasified in H2, at a pressure of approximately 70 bar to produce CH4 via the methanation reaction (1):
Reaction (1) is exothermic and steam would be added to moderate the temperature to approximately 900°C. Next, CH4 is steam-reformed to produce H2 (reaction 2) and the water gas shift reaction (reaction 3) is used to maximise the H2 content of the gas:
Overall, the H2 producing reactions are endothermic. The heat requirement is provided by reaction 1 and by the carbonation of CaO (reaction 4). The latter reaction provides the major portion of the energy and serves to remove the CO2 from the reaction mixture. The resulting shift of the equilibrium in reaction (3) ensures that the H2 concentration is maximised and provides the means to collect a concentrated stream of CO2. Reaction temperatures may be limited by the melting properties of the coal and its mineral matter, the need to avoid the formation of eutectic liquid phases in the solid sorbents used to remove CO2 and the equilibrium controlled inability of CaO to bind CO2 at temperatures above 900°C at a CO2 partial pressure of less than 10 bar. To ensure effective energy utilisation, reactions (2)-(4) need to be conducted in a single reactor. Overall, the scheme results in the formation of 2 extra moles of H2, for each 2 moles H2 used in the methanation stage. The H2 for the methanation is recycled from the H2 stream remaining after the further processing of the gas. The bound CO2 is subsequently released as a concentrated stream for sequestration by heating the CaCO3. Overall, the same amount of CO2 is formed per kg of coal burned with both ZEC and conventional technologies, however, the ZEC process potentially has a higher electrical efficiency (around 65 %), as losses are avoided by the integration of the various process steps. It also isolates the CO2 as a nearly pure stream for storage, by the use of the carbonation/calcination cycle
For people working in this area, please click here.
